- Region:
- Europe
- Category:
- Society
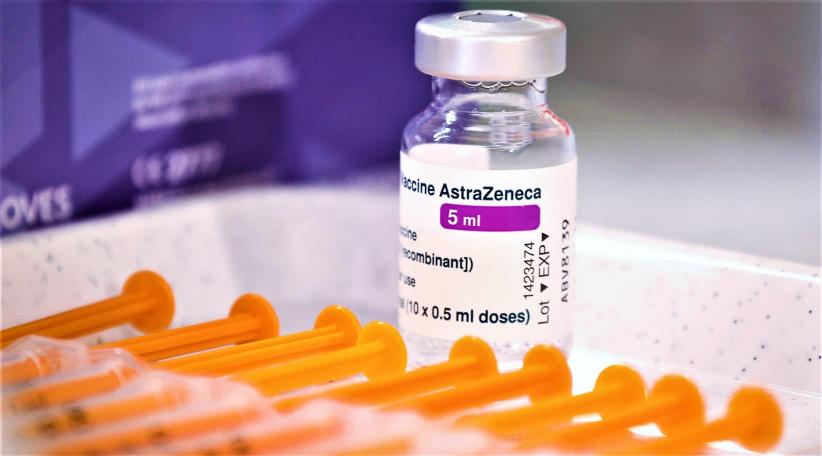
EU drug regulator: Unusual blood clot is 'very rare AstraZeneca side effect'
The EU's medicines regulator says unusual blood clots should be listed as a very rare side effect of the AstraZeneca vaccine for Covid-19.
After a study looking at 86 European cases, the European Medicines Agency (EMA) concluded the benefits of the vaccine outweighed the risk.
The report reflected data on 25 million Europeans administered with the jab.
The EMA could not list specific risk factors such as age or gender, but most blood clot cases were women under 60.
Separately, the UK's vaccine advisory body said that under-30s there were to be offered an alternative jab to AstraZeneca due to the blood clots issue. Some 79 people had suffered rare blood clots after vaccination by the end of March in the UK - 19 of whom had died.
Experts warned of damage to confidence in the UK’s vaccine programme after 10 million adults under 30 were told they will be offered an alternative to the Oxford/AstraZeneca jab following concerns over rare blood clots.
Healthy 18- to 29-year-olds who are not at high risk of Covid should have the option of a different jab if one is available in their area, the government’s joint committee on vaccines and immunisation (JCVI) said, weeks after some European countries suspended the use of Oxford/AstraZeneca jabs in younger people.
For older people, the benefits of the vaccine – the most widely used in the UK – far outweigh the risks, the JCVI added. The UK has recorded 79 rare blood clots cases, 19 of whom died, out of 20m AstraZeneca jabs administered.