European Union authorizes Pfizer's COVID vaccine for kids 5-11
It is the first time the European Medicines Agency has cleared a COVID-19 vaccine for use in young children.
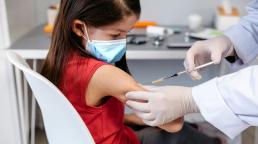
It is the first time the European Medicines Agency has cleared a COVID-19 vaccine for use in young children.
The EU's drugs regulator has said that blood clots should be listed as a "very rare" side effect of the Johnson & Johnson Covid-19 vaccine.
The European Medicines Agency (EMA) on Tuesday recommended a warning should be added to the vaccine’s product information, but stressed that the benefits of the shot – which was put on hold last week in Europe and the US – outweighed its risks.
